Peter Jones, PhD, and Charis Himeda, PhD, found themselves unexpectedly in the media spotlight last week after publication of their paper on the application of CRISPR technology in FSHD cells. In… Read More »
Biology 101 for understanding the CRISPR research
Washington Post features FSH muscular dystrophy story
Today’s Washington Post featured a report on work by a research team that was described in an FSH Society press release. The Post story, “How CRISPR could lead to a cure for… Read More »
Dogs destined to develop muscular dystrophy evade their genetic fate
Golden retrievers with the Duchenne mutation show that disease-modifying genes may provide routes to treat a genetic disorder; possible parallels to facioscapulohumeral muscular dystrophy. Research led by Louis M. Kunkel,… Read More »
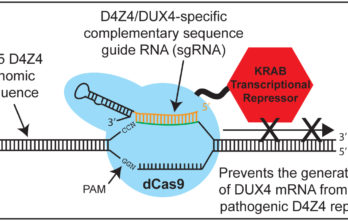
Gene Interference Technology Used Against FSH Muscular Dystrophy Genetic Defect
Proof-of-principle study is the first to use CRISPR technology on the “repeat genome,” as well as its first successful use in primary human muscle cells BOSTON, MA (PRWEB) NOVEMBER 04,… Read More »
International workshop spotlights progress in facioscapulohumeral muscular dystrophy research
Report by Charis Himeda, PhD The FSH Society’s 2015 International Research Consortium and Research Planning Meetings, held in Boston on October 5-6, 2015, brought together over 100 investigators, including leaders… Read More »