Aiming to accelerate disease treatment and care for rare diseases See PaLaDIn Launch Press Release The FSHD Society is embarking on an exciting international collaboration to speed up the development… Read More »
FSHD Society partnering in €21 Million project
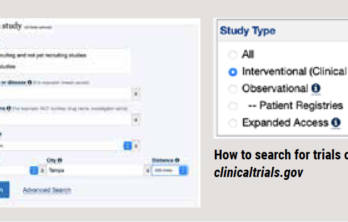
Your practical guide to clinical trials
by Amanda Hill, FSHD Society How do I find clinical trials that are recruiting participants? Our website is one of the best ways to find clinical trials that are recruiting…. Read More »
The children of FSHD – they exist, too
One community – united effort – cure for all by Ally Roets, Tucson, Arizona A few years ago, as we started to hear about clinical trials for promising new drugs… Read More »
Engineering new muscles
Using nature’s scaffolding to coax cells to develop into muscle by Fred Thys, Weymouth, Massachusetts Neil Camarta, who has FSHD and founded the FSHD Canada Foundation, discovered that there is… Read More »
The FACT Alliance
Progress through collaboration by Jamshid Arjomand, PhD, Chief Science Officer As a fan of home improvement shows, I’m fascinated by the evolution of going from an idealized dream home or… Read More »