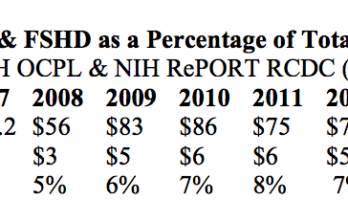[…] Biosciences announced this week that it had received FDA Orphan Drug Designation for its experimental FSHD treatment, called AOC 1020. This does NOT mean the drug is now available for […]
Avidity gets orphan drug designation
Epic Bio takes aim at DUX4
[…] reflect the chance in name from Epic Bio to Epicrispr Biotechnologies. Leading-edge CRISPR biotech chooses FSHD as its first disease target Epicrispr Biotechnologies, based in the San Francisco Bay Area, […]
FSH Society submits testimony to U.S. Congress
[…] Agencies (LHHSE). This year, the Society has requested $24 million FY2017 appropriations for NIH research on FSHD. Identical testimony was submitted to the Senate. The following table from the FSH Society’s testimony […]
Changing mindset about assistive technologies
Emma Weatherley, executive director of Australia’s FSHD Global Research Foundation, has a refreshingly positive outlook on assistive devices. Instead of seeing the need to use a brace or wheelchair as […]
Let’s start a movement!
[…] services I normally use. She even took the time to watch my daughter’s documentary on FSHD, (Lexi Pappas – Growing Up With FSHD) and to visit the FSHD Society website. […]